
A pioneering accreditation initiative in the field of organoid biobanking
Advancing global standards for organoid biobanks in alignment with ISO/IEC 17011 principles
ISO/IEC 17011 aligned (principles-based)
Independent governance structure
International accreditation scope
Organoid-specific standards


About Isoor-AB
ISoOR-AB is an emerging accreditation initiative dedicated to organoid biobanking. It was established within the International Society of Organoid Research (ISoOR) to support the development of quality, consistency, and scientific rigor in this evolving field.
The ISoOR International Standard for Organoid Biobanking (ISoOR-ISOB) provided the technical and normative foundation for the establishment of ISoOR-AB. Operating in alignment with the principles of ISO/IEC 17011, ISoOR-AB directly evaluates and accredits organoid biobanks against the requirements established in the ISoOR-ISOB standard through its own technical and assessment structure.
When appropriate, ISoOR-AB may also utilize Conformity Assessment Bodies (CABs) or external technical experts as complementary support within the evaluation process, while maintaining full authority and final responsibility over accreditation decisions.
The activities of ISoOR-AB include the development of assessment criteria, structured evaluation processes, and mechanisms for verifying ongoing conformity, supporting quality, reliability, and harmonization within the organoid biobanking field.
The governance and operational framework of ISoOR-AB is designed to ensure functional independence in accreditation decision-making. Defined procedures, documentation systems, and conflict-of-interest safeguards are implemented to support impartiality, transparency, and consistency in accreditation activities.
ISoOR-AB aims to contribute to the development of harmonized practices in organoid biobanking and to support the long-term advancement of quality frameworks in this emerging field.


Operating in accordance with ISO/IEC 17011 principles, ISoOR-AB ensures impartial, transparent, and globally recognized accreditation of organoid biobanks.
Our Services
Accrediting organoid biobanks with trusted standards.
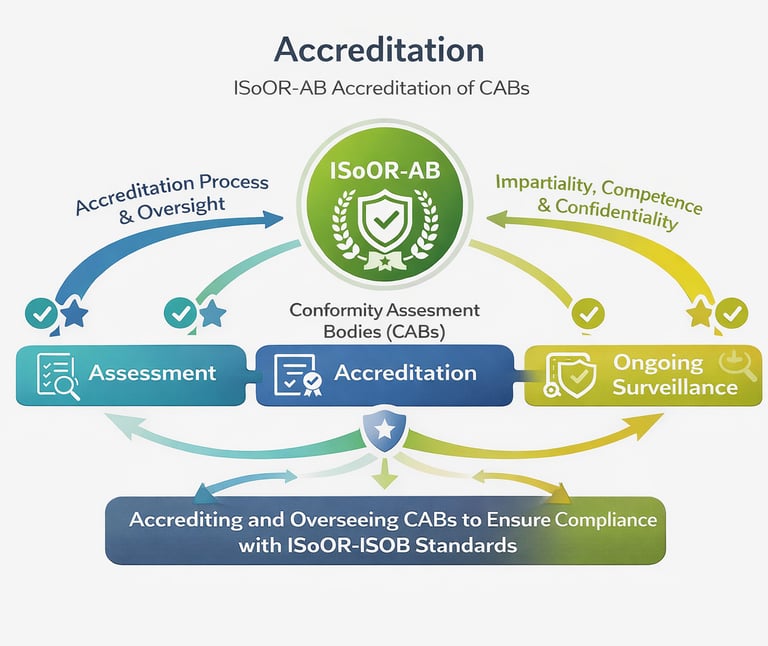
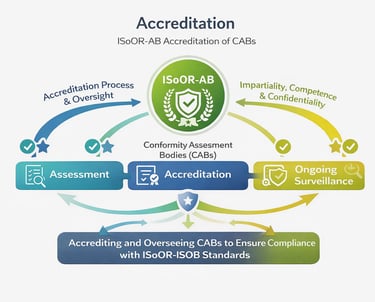
The ISoOR-AB accreditation process ensures that organoid biobanks are competent to operate in accordance with the ISoOR-ISOB standard. It involves a thorough evaluation of each biobank’s policies, procedures, technical capabilities, and resources, followed by on-site or remote assessments of operational practices and personnel qualifications.
Accreditation decisions are made by an impartial ISoOR-AB committee, ensuring transparency and objectivity. Where applicable, external conformity assessment outputs may be considered as supporting evidence within the evaluation process. Accredited biobanks are subject to periodic reassessment to ensure continued compliance and to maintain high standards of quality, integrity, and scientific rigor.
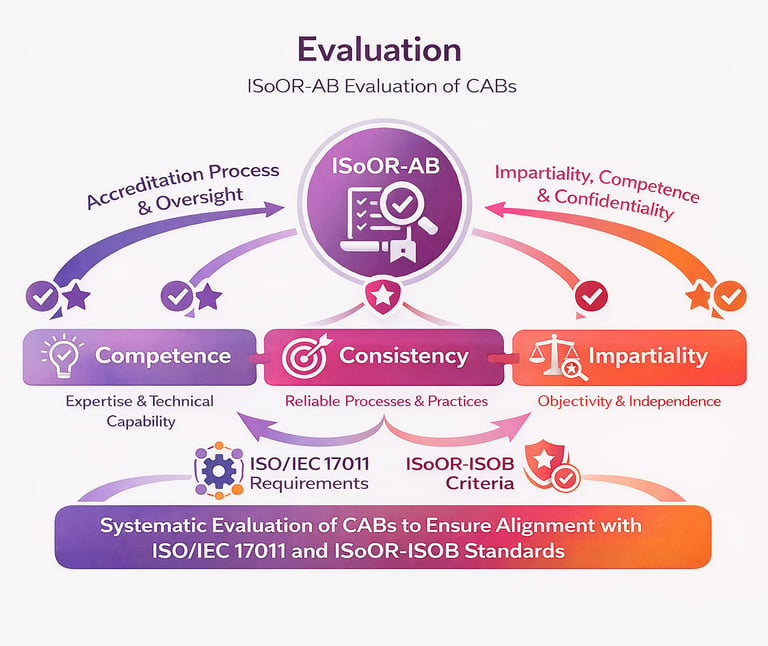
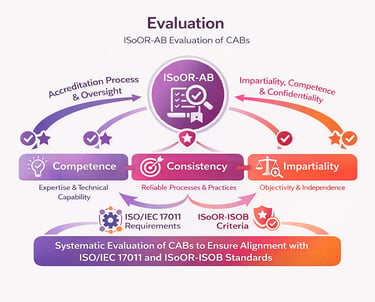
ISoOR-AB conducts comprehensive and systematic evaluations of organoid biobanks to assess their competence, consistency, and impartiality in accordance with the ISoOR-ISOB standard. These evaluations include detailed reviews of institutional procedures, documentation, technical expertise, and personnel qualifications to ensure compliance with established requirements.
Assessment activities may involve on-site or remote evaluations, interviews, and sampling of operational records to verify adherence to principles inspired by ISO/IEC 17011 and international best practices. Findings are analyzed by an impartial evaluation team, and recommendations are issued to address any identified gaps. Through this process, ISoOR-AB ensures that accredited biobanks operate with high levels of objectivity, reliability, and scientific rigor, maintaining confidence in organoid research and biobanking practices worldwide.
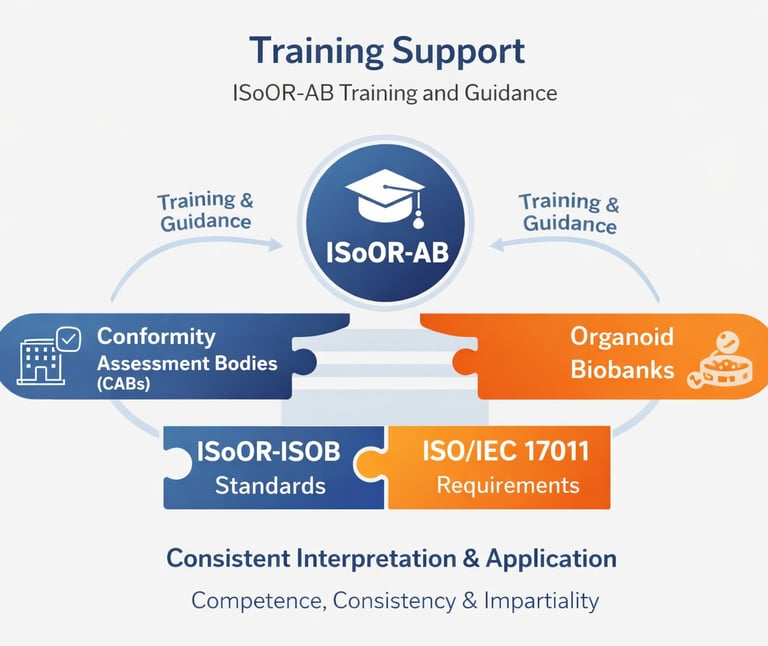
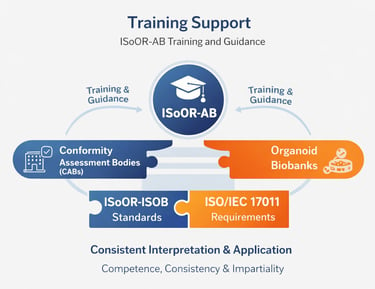
ISoOR-AB provides structured training and guidance to organoid biobanks, as well as to CABs where applicable, to ensure consistent and accurate interpretation and application of the ISoOR-ISOB standard. Training programs cover key aspects of the standard, including quality management, sample handling, data integrity, and ethical compliance, while emphasizing competence, impartiality, and scientific rigor.
These programs combine workshops, practical exercises, and expert-led guidance to support the implementation of best practices in biobank operations and, where relevant, conformity assessment activities. By reinforcing knowledge, skills, and understanding of the principles of ISO/IEC 17011 and international best practices, ISoOR-AB promotes consistency, transparency, and high professional standards, fostering confidence in the accreditation system across the organoid research community.
For assistance with training, reach out to us at info@isoor-ab.org
Accreditation Process
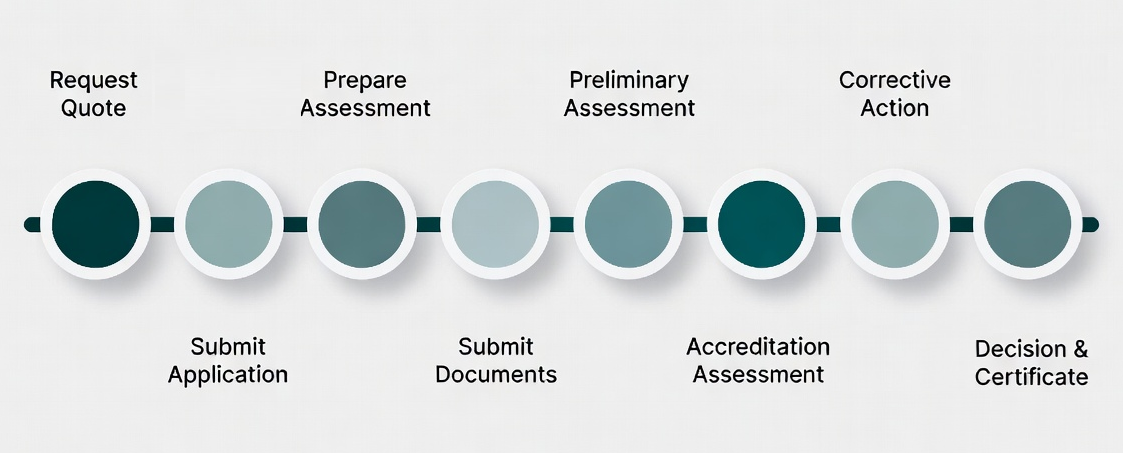
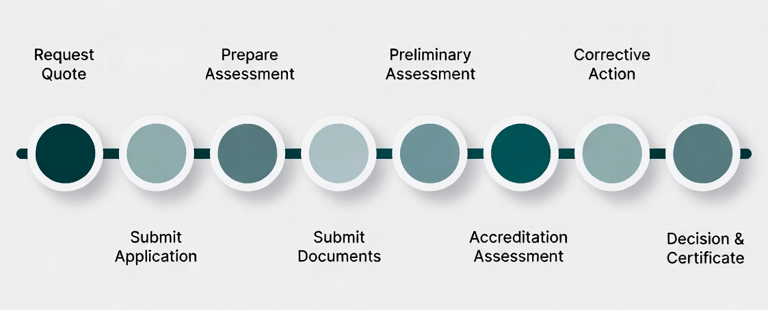
1- Required Documents
Quality manual and operational procedures
Personnel qualifications and training records
Scope of activities and biosafety documentation
2- Complete the Application Forms
3- Attach Supporting Documentation
Fill out the relevant application documents listed in Step 1 with accurate information about your organoid biobank, including its scope of accreditation and quality management system
4- Submit your Application
Submit the completed application and supporting documents via the ISoOR-AB portal or email to info@isoor-ab.org. Pay the application fee as applicable
5-Assessment by ISoOR-AB Expert Team
ISoOR-AB conducts a direct assessment of organoid biobanks against the ISoOR-ISOB standard through document review, interviews, and on-site or remote evaluations. Where available, CAB reports may be used as supporting evidence, but the final evaluation is carried out independently by ISoOR-AB experts. Corrective actions may be requested if needed, and accreditation is granted once all requirements are met.
ISoOR-AB reviews the assessment findings and may request corrective actions if needed. Once all requirements are met, ISoOR-AB makes the accreditation decision and issues the accreditation certificate.
6- Accreditation Decission
Organizational Structure
&
Governance Framework
The organizational structure of ISoOR-AB is designed to ensure clear allocation of responsibilities, transparency in decision-making, and effective operational control. A well-defined governance framework is essential to maintain consistency, accountability, and impartiality across all accreditation activities.
In alignment with ISO/IEC 17011, ISoOR-AB establishes independent committees, documented procedures, and defined roles that prevent conflicts of interest and safeguard objective evaluations. This structure supports the separation of authority between governance, operational, and decision-making functions, reinforcing credibility and trust in the accreditation process.
It also ensures that ISoOR-AB operates independently from its founding body, International Society of Organoid Research, thereby upholding the principles of impartiality and integrity required by international standards.
Together, the organizational structure and governance framework provide the foundation for a robust, transparent, and internationally recognized accreditation system.er, the organizational structure and governance framework provide the foundation for a robust, transparent, and internationally recognized accreditation system.


Governance and Independence Framework Documentation (Aligned with ISO/IEC 17011 Principles)
ISoOR-AB Accreditation Policy
The ISoOR-AB organizational structure defines roles, reporting lines, and functional separation mechanisms to support impartiality, independence, and integrity in the accreditation process. It is designed in accordance with the principles of ISO/IEC 17011 and international best practices for peer-based accreditation systems, ensuring a clear distinction between ISoOR-AB’s accreditation functions and the standard-setting activities of founding organization (ISoOR).
ISoOR-AB Governance and Decision-Making
ISoOR-AB maintains a transparent governance framework ensuring impartial, consistent accreditation of organoid biobanks. Its governance structure is organized into functional blocks, including the Governance Board, Operational Management, Technical Assessment Function, Impartiality and Ethics Function, and Appeals and Review Function. Accreditation decisions are based on direct assessment of organoid biobanks by qualified expert teams within the Technical Assessment Function, with oversight provided by the Governance Board and safeguards for impartiality ensured through the Impartiality and Ethics Function. External conformity assessment evidence may be considered where appropriate to support decision-making. Appeals are handled through an independent review mechanism to ensure fairness, transparency, and procedural integrity while maintaining confidentiality.
ISoOR-AB Committee Governance Charter
ISoOR, the parent and founding organization of ISoOR-AB, advances organoid research and established ISoOR-ISOB, the first international standard for organoid biobanking (March 2025). Operating as an independent accreditation body, ISoOR-AB directly accredits organoid biobanks against this standard, ensuring quality, impartiality, and consistency through governance structures and procedures informed by the principles of ISO/IEC 17011, robust peer-review processes, and engagement with international stakeholders.
ISoOR-AB Impartiallity Policy
The ISoOR-AB Impartiality Policy defines how ISoOR-AB maintains unbiased, independent, and transparent accreditation operations. It establishes requirements for identifying and managing conflicts of interest, structuring committees to ensure balanced and independent decision-making, conducting accreditation assessments based on objective evidence and expert peer review, and safeguarding confidentiality. The policy is guided by the principles of ISO/IEC 17011 and international best practices for peer-based accreditation systems.
Legal Status & Ownership
ISoOR-AB is an independent, non-profit legal entity. It was founded by the International Society of Organoid Research (ISoOR) but operates as a standalone organization with its own legal identity, governance structure, and decision-making authority.
ISoOR-AB has the legal capacity to enter into contracts, employ staff, and conduct accreditation activities for Conformity Assessment Bodies (CABs) or organoid biobanks in accordance with its mandate.
While ISoOR-AB maintains operational and decision-making independence from ISoOR, it retains a historical and institutional founding relationship with ISoOR, without implying legal control or ownership by ISoOR.
The Board of Directors is responsible for ensuring compliance with legal obligations, maintaining non-profit status, and safeguarding the independence and integrity of the accreditation system.
ISoOR-AB and ISoOR: Independence and Complementarity
The ISoOR-AB Accreditation Policy defines a private, voluntary accreditation framework under which ISoOR-AB directly evaluates organoid biobanks through its appointed expert assessors and peer-review mechanisms, similar to established international models.
In support of its accreditation decision-making process, ISoOR-AB may, where appropriate, consider conformity assessment outputs generated by Conformity Assessment Bodies (CABs) or other qualified third-party evaluations as supplementary evidence. Such inputs serve to inform and strengthen the accreditation assessment but do not replace or delegate ISoOR-AB’s independent evaluation and final accreditation decision.
The system is aligned with the principles of ISO/IEC 17011, particularly impartiality, competence, consistency, and transparency, while operating independently of national accreditation systems
Accreditation Framework
Defines the principles, structure, and processes that underpin the ISoOR-AB accreditation system. Ensures transparency and clarity for stakeholders and provides CABs with context for operational requirements.
ISoOR-AB Accreditation Process Procedure
This document outlines how ISoOR-AB evaluates and accredits organoid biobanks. It describes each stage of the accreditation process—from application, document review, and on-site assessment, to nonconformity management, decision-making, surveillance, and re-accreditation. The procedure ensures transparent, impartial, and consistent accreditation practices, providing stakeholders with confidence in the competence, quality, and reliability of accredited biobanks. Where applicable, ISoOR-AB may also consider conformity assessment outputs from qualified third-party bodies as supporting evidence within the evaluation process.
For Stakeholders:
For Conformity Assessment Bodies (CABs):
ISoOR-AB Conflict of Interest and Recusal Policy establishes a framework to identify, disclose, manage, and mitigate conflicts of interest in all accreditation activities. It applies to governance members, staff, technical experts, auditors, and external advisors involved in the direct accreditation of organoid biobanks. The policy defines conflicts of interest as any actual or perceived personal, professional, financial, or institutional interest that could influence or appear to influence impartial decision-making. All participants are required to submit and update declarations of interest and disclose conflicts prior to participation in assessments or decisions. Where conflicts arise, ISoOR-AB applies proportionate measures including disclosure, restriction of access, recusal from deliberations and decisions, reassignment of duties, or removal from roles in severe cases, with all recusal actions formally recorded. Oversight is provided through governance and impartiality functions to ensure integrity, independence, and procedural fairness in accordance with ISO/IEC 17011 principles and international best practices.
Support & Accountability
ISoOR-AB Appeals and Complaints Procedure
ISoOR-AB provides impartial procedures for handling complaints and appeals from accredited organoid biobanks, applicants, and relevant stakeholders. Concerns may be submitted via email or an online form and are reviewed by designated independent panels. Decisions are communicated transparently, and confidentiality is strictly maintained throughout the process. Where necessary, corrective actions are implemented to ensure fairness, accountability, and continuous improvement. The procedure is guided by the principles of ISO/IEC 17011 and international best practices for peer-based accreditation systems.
ISoOR-AB Conflict of Interest and Recusal Policy
Stakeholder Engagement
As part of its international development strategy, ISoOR-AB is establishing a structured stakeholder engagement program to strengthen global alignment, transparency, and credibility in organoid biobanking accreditation.
ISoOR-AB actively engages with regulators, accreditation bodies, research institutions, and industry partners to foster a collaborative and open ecosystem. This initiative ensures that the accreditation framework reflects internationally recognized best practices, supports harmonization across regions, and builds mutual trust among stakeholders.
We welcome the participation of all relevant stakeholders—including regulators, researchers, industry partners, and accreditation bodies—in contributing to this initiative. Your expertise and perspectives are essential to ensuring that ISoOR-AB’s accreditation system remains robust, reliable, and globally relevant.
Together, we aim to advance a transparent, harmonized, and trusted framework for organoid biobanking worldwide.


FAQ
What is ISOOR-AB?
ISoOR-AB is the accreditation body of the International Society for Organoid Research (ISoOR). It operates based on the principles of ISO/IEC 17011, which establish best practices for accreditation bodies. ISoOR-AB ensures that the organoid biobanks it evaluates comply with high standards of quality, transparency, and integrity, providing credibility and confidence in organoid research and applications at an international level.
Why does ISoOR-AB adopt the principles of ISO/IEC 17011 in its accreditation system?
Alignment with the principles of ISO/IEC 17011 enables ISoOR-AB to adopt consistent, transparent, and impartial accreditation practices. This international standard establishes best practices for accreditation bodies, helping to strengthen the credibility, reliability, and integrity of the accreditation system. By following these principles, ISoOR-AB fosters trust among laboratories, researchers, and other stakeholders in the field of organoid research.
Where can I find the documents?
All governance documents are available here as downloadable PDFs for easy reference.
Who uses these documents?
These documents are used by accreditation bodies and auditors to ensure compliance with international standards. They are also used by organoid biobank applicants to prepare their applications, demonstrate adherence to requirements, and align their practices with globally recognized standards.
Are updates frequent?
Updates occur as standards evolve to maintain alignment.
How do I stay informed about changes?
Subscribe to notifications or check back regularly for the latest documents.
Why align with ISoOR‑ISOB?
Alignment with ISoOR‑ISOB ensures that ISoOR-AB’s accreditation process is specifically tailored to organoid biobanking and research. It provides a clear framework of best practices, technical requirements, and evaluation criteria for applicants, promoting consistency, transparency, and high-quality standards in organoid biobanks worldwide.


